|
However, if the polypeptide adopts its correct shape, it can then transit to the Golgi and advance further through the secretory pathway. In this process, misfolded protein is retro-translocated across the ER membrane into the cytosol, where it is ubiquitylated and targeted for degradation via the 26S proteasome. These alterations to the glycan chain help to monitor the folding status of the nascent polypeptide and act as an important quality control measure ( Wang and Kaufman, 2016 Hetz and Papa, 2017).Ī failure in the polypeptide chain to adopt its native conformation may lead to activation of degradation pathways, including ER-associated degradation (ERAD) ( Hampton, 2002). 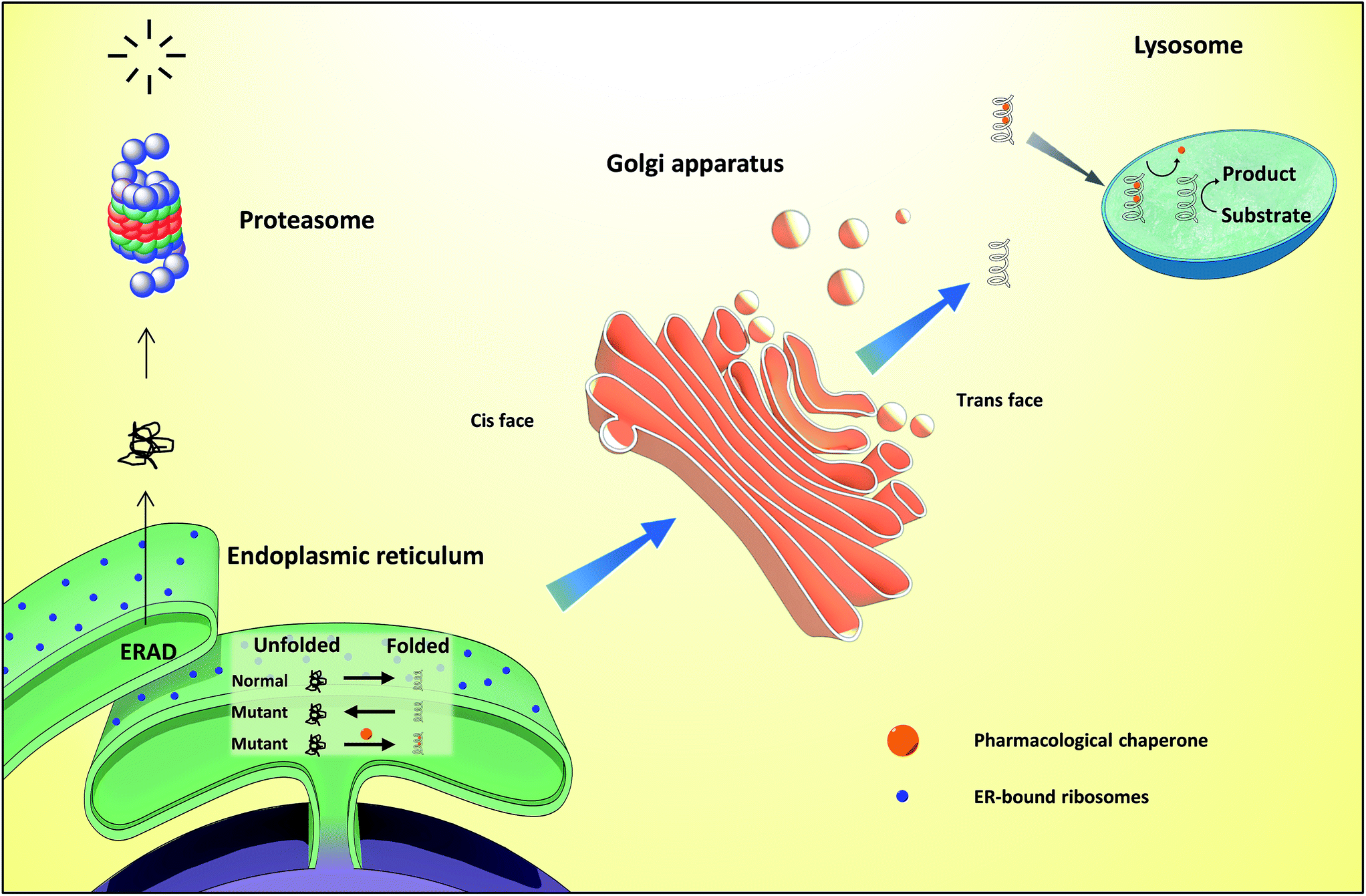
Once in the ER, there are a number of glycosylating enzymes that either trim or add to the core N-linked oligosaccharide depending on the progress of protein folding. Another essential modification is the attachment of N-linked oligosaccharides to the nascent chain, which occurs upon entry into the ER. This bond is important for maintaining tertiary and quaternary protein structure and is catalyzed by protein disulphide isomerase (PDI). Post-translational modifications are also critical for correct protein folding and one such modification is disulphide bond formation. BiP binds to nascent polypeptide chains to prevent their aggregation initially and subsequently, facilitates their folding in order for the polypeptide to achieve its native conformation. One of the most abundant proteins within the ER is the Hsp70-type chaperone, BiP (binding-immunoglobulin protein aka GRP-78). There are various ER resident enzymes and chaperones that increase the efficiency of protein folding of the nascent polypeptide. Once inside the ER, the signal sequence is cleaved by signal peptidase and the translocated polypeptide undergoes post-translational modification and chaperone-assisted folding to help it to form its correct three-dimensional shape. The newly translated polypeptide contains an N-terminal signal sequence that is recognized by signal recognition particle (SRP), which enables its insertion into the ER via the transolocon complex. Proteins that reside in the ER, along with proteins destined for the Golgi, plasma membrane, and extracellular space are synthesized in ribosomes that are attached to the ER membrane. The endoplasmic reticulum is a major site for protein folding and maturation within the eukaryotic cell. We draw a comparison between the recently proposed allosteric model for UPR induction and the role of Hsp70 during polypeptide import to the mitochondrial matrix.  
Here, we discuss the structural and molecular basis of IRE1 stress signaling, with particular focus on novel mechanistic advances. The key UPR signal activator, IRE1, responds to stress by propagating the UPR signal from the ER to the cytosol. However, the physiological demands or aberrations in folding may result in an imbalance which can lead to the accumulation of misfolded protein, also known as “ER stress.” The unfolded protein response (UPR) is a cell-signaling system that readjusts ER folding capacity to restore protein homeostasis. The cellular requirement to synthesize proteins within the ER is matched by its folding capacity. The endoplasmic reticulum (ER) is an important site for protein folding and maturation in eukaryotes. Department of Life Sciences, Imperial College London, London, United Kingdom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
